Tag Archive for: research
 https://www.college.ucla.edu/wp-content/uploads/2025/10/NewsroomMainImage_Final003-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-10-16 09:06:372025-10-20 09:08:54How two Bruins used film and science to reframe a ‘spooky’ species
https://www.college.ucla.edu/wp-content/uploads/2025/10/NewsroomMainImage_Final003-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-10-16 09:06:372025-10-20 09:08:54How two Bruins used film and science to reframe a ‘spooky’ species
‘The future of food’
UCLA experts discuss how we move this ‘universal connector’…
 https://www.college.ucla.edu/wp-content/uploads/2025/10/Ramsdell-Hero-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-10-06 11:04:332025-10-13 11:25:38UCLA alumnus Fred Ramsdell wins 2025 Nobel Prize in physiology or medicine
https://www.college.ucla.edu/wp-content/uploads/2025/10/Ramsdell-Hero-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-10-06 11:04:332025-10-13 11:25:38UCLA alumnus Fred Ramsdell wins 2025 Nobel Prize in physiology or medicine https://www.college.ucla.edu/wp-content/uploads/2025/10/sealion-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-10-03 11:04:342025-10-13 11:25:46L.A. Times: Marine mammals are dying in record numbers along the California coast
https://www.college.ucla.edu/wp-content/uploads/2025/10/sealion-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-10-03 11:04:342025-10-13 11:25:46L.A. Times: Marine mammals are dying in record numbers along the California coast https://www.college.ucla.edu/wp-content/uploads/2025/10/MeetingMinds_DavidMeyersQA_363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-28 10:44:232025-10-13 10:50:37UCLA’s David Myers on how the public university can address society’s ‘kindness deficit’
https://www.college.ucla.edu/wp-content/uploads/2025/10/MeetingMinds_DavidMeyersQA_363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-28 10:44:232025-10-13 10:50:37UCLA’s David Myers on how the public university can address society’s ‘kindness deficit’ https://www.college.ucla.edu/wp-content/uploads/2025/10/Frenk-LA-Times-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-26 10:44:182025-10-13 10:50:42UCLA chancellor ready to stand firm against Trump demands, unless they’re ‘valid’
https://www.college.ucla.edu/wp-content/uploads/2025/10/Frenk-LA-Times-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-26 10:44:182025-10-13 10:50:42UCLA chancellor ready to stand firm against Trump demands, unless they’re ‘valid’ https://www.college.ucla.edu/wp-content/uploads/2025/10/morning_658e91aa-25dc-4e43-8937-39856a7d2ce7-prv-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-24 10:22:402025-10-13 10:27:22UCLA anthropologists uncover immune roots of morning sickness
https://www.college.ucla.edu/wp-content/uploads/2025/10/morning_658e91aa-25dc-4e43-8937-39856a7d2ce7-prv-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-24 10:22:402025-10-13 10:27:22UCLA anthropologists uncover immune roots of morning sickness https://www.college.ucla.edu/wp-content/uploads/2025/10/Newsroom-ShapiroHomesick-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-24 10:22:392025-10-13 10:27:30‘Homesick’: Nicholas Shapiro on finding our way from toxic homes to healthier futures
https://www.college.ucla.edu/wp-content/uploads/2025/10/Newsroom-ShapiroHomesick-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-24 10:22:392025-10-13 10:27:30‘Homesick’: Nicholas Shapiro on finding our way from toxic homes to healthier futures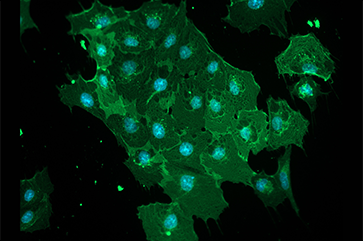 https://www.college.ucla.edu/wp-content/uploads/2025/10/pablo-cell-image-newsroom-size-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-23 10:22:402025-10-13 10:27:25Headspace invaders: How mosquito-borne viruses breach the brain’s defenses
https://www.college.ucla.edu/wp-content/uploads/2025/10/pablo-cell-image-newsroom-size-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-23 10:22:402025-10-13 10:27:25Headspace invaders: How mosquito-borne viruses breach the brain’s defenses https://www.college.ucla.edu/wp-content/uploads/2025/10/abigail-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-18 09:23:062025-10-13 09:26:28How to help students have difficult conversations in the classroom
https://www.college.ucla.edu/wp-content/uploads/2025/10/abigail-363-241.png
241
363
Alvaro Castillo
https://www.college.ucla.edu/wp-content/uploads/2019/07/Uxd_Blk_College-e1557344896161.png
Alvaro Castillo2025-09-18 09:23:062025-10-13 09:26:28How to help students have difficult conversations in the classroom
